The ret proto-oncogene products (proto-Ret protein) are expressed as 150kDa and 170kDa glycoproteins in neuroblastoma cells and as 150kDa and 190kDa glycoproteins in leukemia cells. These proteins are produced from a single polypeptide of 120kDa by posttranslational glycosylation. Although expression of the ret proto-oncogene was frequently detected in human tumors such as neuroblastoma, pheochromocytoma and thyroid medullary carcinoma, its physiological function is unknown. It turned out that the extracellular domain of the proto-Ret protein contains a cadherin-related sequence that is known to be important for Ca2+-dependent homophilic binding of cadherins. The homologous sequence found in the proto-Ret protein consists of about 110 amino acids and is tandemly repeated 3 - 4 times in the extracellular domains of all vertebrate cadherins. The sequence of the proto-Ret protein showed 20-30% identity with the member of the cadherin superfamily in the amino acid level. This suggests that possibility that the proto-Ret protein may function as a cell adhesion molecule like cadherins. For research use only, not for use in diagnostic procedures.
- application:
- WB, IP
- Catalog number:
- 18128
- Datasheet:
- formulation:
- Lyophilized product from 1% BSA in PBS containing 0.05% NaN3
- immunogen:
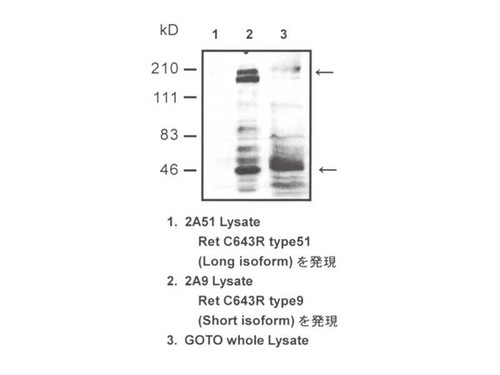
- Synthetic peptide for C-Terminal of Human c-Ret Long Isoform (ANWMLSPSAAKLMDTFDS)
- MSDS:
- notes:
- For research use only, not for use in diagnostic procedures.
The datasheet for this product (see above) is intended to serve as an example only. Please refer to the datasheet provided with the antibody for precise details. - Other names:
- Please see datasheet
- Protocol:
- purification:
- Affinity Purified with synthetic peptide
- size:
- 200 µg
- specificity:
- Long Isoform specific
- storage:
- Lyophilized product, 5 years at 2 - 8 °C; Solution, 2 years at -20 °C.
- Species:
- Human
- Host:
- Rabbit