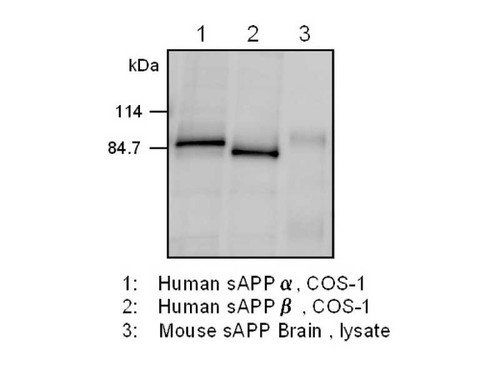
Amyloid precursor protein (APP) is precursor protein of Amyloid β which is major constituent of senile plaque in Alzheimer's disease. It is known that there are three major isoforms, APP695, APP751 and APP770, and are generated from alternative splicing of common precursor mRNA. Processing of APP occurs by two major pathways, non-amyloidogenic pathway and amyloidogenic pathway. The non-amyloidogenic pathway is mediated by α and γ-secretases and gives rise to a large fragment known as soluble APPα (sAPPα) and a small 3 kDa peptide known as p3. On the other hand, the Amyloidogenic pathway is mediated by β- and γ-secretases and yields soluble APPβ (sAPPβ) and Amyloid β. The physiologic function of APP itself is not clear, however, it is supposed that the function of APP in neuron system is different from that in other organ. Amyloidβ is derived by the sequential cleavage of amyloid precursor protein (APP) by beta- and gamma-secretases. A double missense mutation (Lys670→Asn and Met671→Leu) in APP found in a Swedish pedigree (APPβ-sw) elevates Abeta40 and Abeta42 production, and the mutation is utilized in establishment of transgenic mice overexpress a mutant form of human amyloid precursor protein. Amyloidβproduction and, beta-secretase cleavage of APPβ-sw reportedly occur in the endoplasmic reticulum (ER), Golgi and endocytic compartments. In nerve cells, APP containing an N- or O-type sugar chain modification (mature APP) is phosphorylated at the Thr668 position (APP695) by the actions of Cdk5 and c-Jun NH2-terminal kinase (JNK), which are nerve-specifically activated, and becomes translocated to the cell membrane and neuritis. It has been considered that the phosphorylation induces structural changes in the cytoplasmic domain of APP and influences Aβ production. Regulation of the bonding of APP with FE65 is believed to be involved in information transmission. For research use only, not for use in diagnostic procedures.
- application:
- WB, IP
- Catalog number:
- 28021-S
- Datasheet:
- formulation:
- Lyophilized product from 1% BSA in PBS containing 0.05% NaN3
- immunogen:
- Synthetic peptide of Phosphorylated part of APP (DAAVpTPEE)
- MSDS:
- notes:
- For research use only, not for use in diagnostic procedures.
The datasheet for this product (see above) is intended to serve as an example only. Please refer to the datasheet provided with the antibody for precise details. - Protocol:
- purification:
- Purified with antigen peptide
- size:
- 10 µg
- specificity:
- Reacts with human and mouse, and not reacts with non-phosphorylated APP
- storage:
- Lyophilized product, 5 years at 2 - 8 °C; Solution, 2 years at -20 °C.
- Host:
- Rabbit
- Additional info: