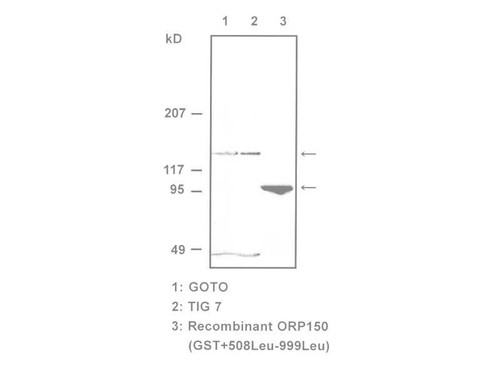
Heat shock proteins (HSPs)/stress proteins are molecular chaperones that are induced by various environmental and physiological stimuli. The 150-kDa oxygen-regulated protein (ORP150), a new member of HSP family, functions as a molecular chaperone in the endoplasmic reticulum (1). It is reported that the ORP150 is an integral participant in ischemic cytoprotective pathways. (2). And, the ORP150 is expressed in human wounds along with VEGF. Colocalization of these two molecules was observed in macrophages in the neovasculature, suggesting a role of ORP150 in the promotion of angiogenesis (3). Furthermore, ORP150 is up-regulated in tumors and, in breast tumors, may be associated with tumor invasiveness. For research use only, not for use in diagnostic procedures.
- application:
- IHC, WB, IP
- Catalog number:
- 10301
- clone:
- 2F07
- concentration:
- Please see datasheet
- Datasheet:
- formulation:
- Lyophilized product from 1% BSA in PBS containing 0.05% NaN3
- immunogen:
- Recombinant Protein (GST-ORP150 (Leu 508 - Leu 999))
- isotype:
- IgG2a
- MSDS:
- notes:
- For research use only, not for use in diagnostic procedures.
The datasheet for this product (see above) is intended to serve as an example only. Please refer to the datasheet provided with the antibody for precise details. - Other names:
- Please see datasheet
- Protocol:
- size:
- 100 µg
- storage:
- Lyophilized product, 5 years at 2 - 8 °C; Solution, 2 years at -20 °C
- Species:
- Human
- Host:
- Mouse
- References: