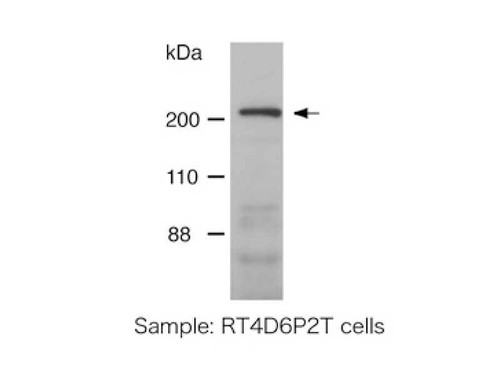
Synaptotagmin family proteins consist of a single N-terminal transmembrane domain and C-terminal tandem C2 domains (C2A and C2B), which have the ability to bind Ca2+ and phospholipid, and 15 synaptotagmin isoforms have been reported in the mouse and humans to date. The best-characterized isoform, synaptotagmin I, is essential for synaptic vesicle exocytosis and endocytosis. It has been reported that i) synaptotagmin IV expression is regulated by neural activity and that it is thought to be involved in synaptic plasticity, ii) that synaptotagmin IV is present on dense-core vesicles in NGF-differentiated PC12 cells and may control their exocytosis, and iii) that synaptotagmin IV regulates glutamate release by astrocytes. For research use only, not for use in diagnostic procedures.
- application:
- ICC, WB, IP
- Catalog number:
- 18977
- Datasheet:
- formulation:
- Lyophilized product from 1% BSA in PBS containing 0.05% NaN3
- immunogen:
- Recombinant Synaptotagmin IV C2A domain
- MSDS:
- notes:
- For research use only, not for use in diagnostic procedures.
The datasheet for this product (see above) is intended to serve as an example only. Please refer to the datasheet provided with the antibody for precise details. - Other names:
- Please see datasheet
- Protocol:
- purification:
- Affinity Purified with antigen protein
- size:
- 100 µg
- specificity:
- Cross-react with mouse and rat Synaptotagmin IV. Not cross-react with the other members of synaptotagmin family
- storage:
- Lyophilized product, 5 years at 2 - 8 °C, Solution, 2 years at -20 °C
- Host:
- Rabbit
- Additional info: